On the basis of his model, Rutherford was able to predict the size of the nucleus. Very few α-particles, those that collide with the nucleus, would face a rebound. Only those particles that come in the close vicinity of the positively charged nucleus get deviated from their path. The α particles passing through the atom in the region of the electrons would pass straight without any deflection. On the basis of the proposed model, the experimental observations in the scattering experiment could be explained. Schematic Figure shows Rutherford’s Experimentįor free demos and animated video lectures, click on NCERT Class 11 Chemistry. The rest of an atom must be empty space which contains the much smaller and negatively charged electrons. According to Rutherford’s model, an atom contains a dense and positively charged region located at its center it was called the nucleus, all the positive charge of an atom, and most of its mass was contained in the nucleus. The results of α-ray scattering experiment were explained by Rutherford in 1911 and another model of the atom was proposed. (iv) About 1 in every 12000 particles experienced a rebound. (iii) A few particles were deflected by large angles. (ii) Some of the α-particles were deflected by small angles. (i) Most of the α-particles passed straight through the gold foil. But the actual results of the experiment were quite surprising. According to Thomson’s model, it was expected that the alpha particles would just pass straight through the gold foil and could be detected by a photographic plate placed behind the foil. In this experiment, a stream of α particle from a radioactive source was directed on a thin (about 0.00004 cm thick) piece of gold foil. This led to the failure of Thomson’s model of the atom. 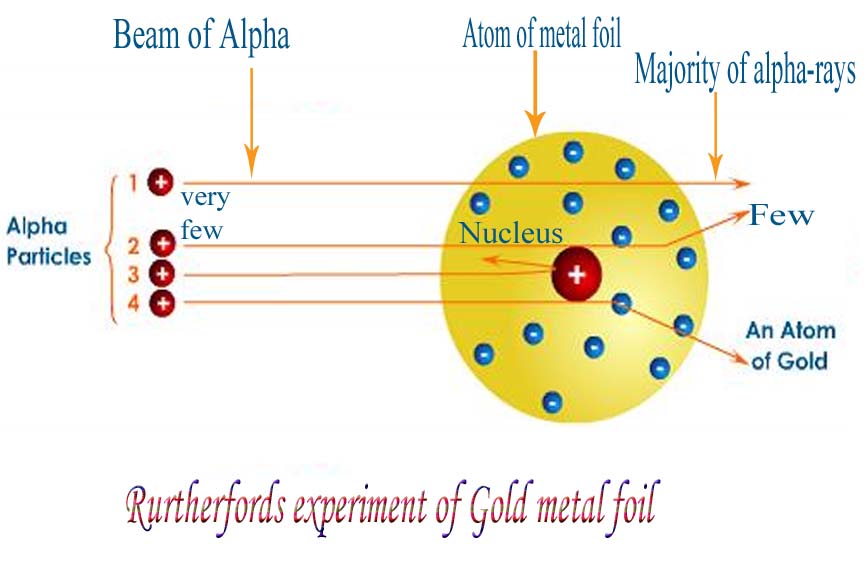
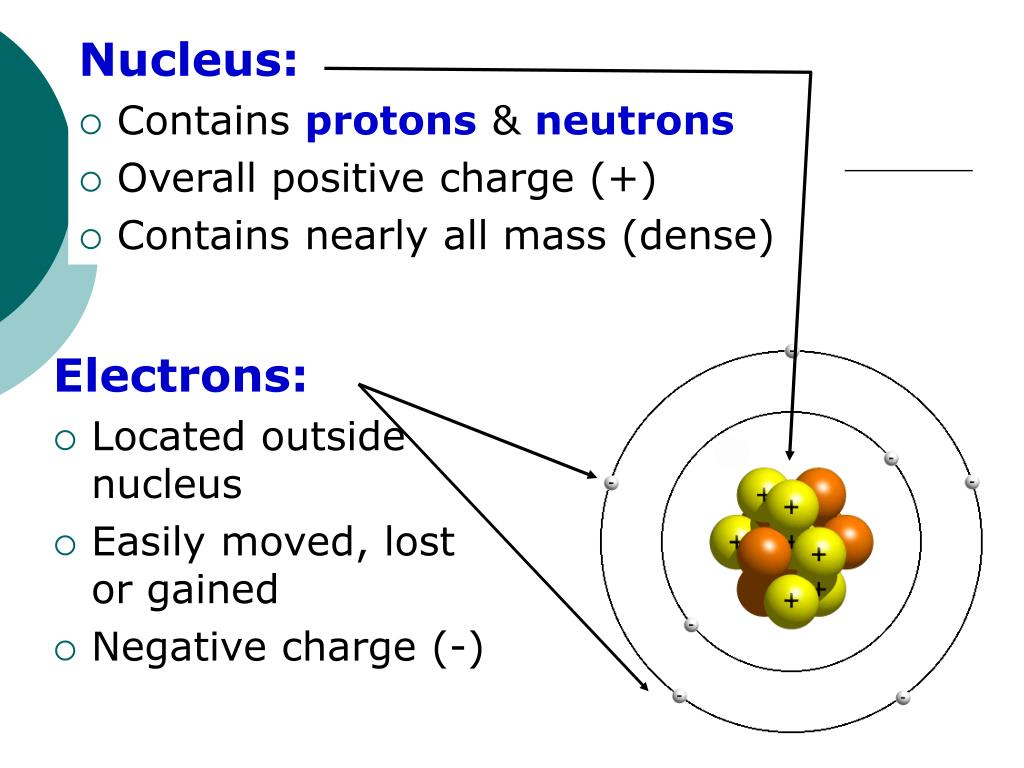
In 1910, Hans Geiger (Rutherford’s technician) and Ernest Marsden ( Rutherford’s student) performed the famous α-ray scattering experiment. The alpha particles are helium nuclei, which can be obtained by the removal of two electrons from the helium atom. They were studying the effect of alpha (α) particles on the matter. Rutherford Model Of Atom Class 11 Chemistry ( Rutherford’s Atomic Model)Įrnest Rutherford and his co-workers were working in the area of radioactivity. Class 11 Chemistry – Structure of Atom Structure of Atom – Rutherford’s Atomic Model
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
